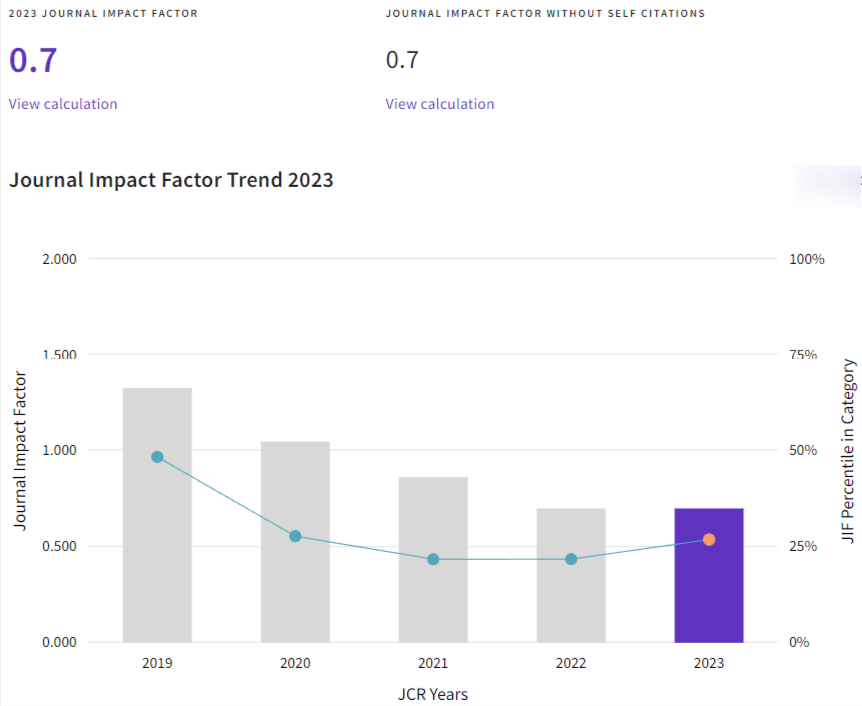
Insulin-like growth factor 1 (IGF-1) concentrations in synovial fluid of sound and osteoarthritic horses, and its correlation with proinflammatory cytokines IL-6 and TNF
DOI:
https://doi.org/10.22319/rmcp.v14i1.6241Palabras clave:
Insulin growth factor 1 (IGF-1), Horse, Osteoarthritis (OA), LameResumen
Insulin-like growth factor I (IGF-1) is the most important known growth factor for cartilage repair in horses. It promotes mitosis of chondrocytes, collagen II expression, and extra cellular matrix production. Osteoarthritis (OA) is the most common musculoskeletal condition that causes lameness and poor performance in sport horses. A total of 11 lame horses were clinically and radiographically evaluated, and all were confirmed to suffer a front metacarpophalangeal lameness by a positive flexion test, a low-4-point nerve block and an intraarticular block. Total protein, IGF-1, IL-6 and TNFa were determined by ELISA, demonstrating changes and different correlations between clinical condition, radiographic changes and degree of inflammation. All horses with joint associated pain and therefore associated lameness, demonstrated a significant increase of total protein (P<0.0001) and IGF-1 concentration (P<0.05). Concentrations of IL-6 and TNFa between controls and lame horses demonstrated significant differences (P<0.01 and P<0.001 respectively). Horses with less radiographic changes, demonstrated the highest IGF-1 expression in synovial fluid, and horses with more chronic OA conditions had very similar IGF-1 expression levels than control joints. In all lame joints, it was identified by Western blot a lighter isoform of IGF-1 (~7.5 kDa) which was inflammation related and it is the molecular weight of the mature peptide, and all control joints expressed a heavier isoform (~12 kDa). This finding could lead to new research for sequencing and targeting the isoform which is not expressed during an inflammatory process within a joint, and to have a better understanding of its role in the horse’s joint.
Descargas
Citas
Pfeffer LA, Brisson BK, Hanquin L, Barton ER. The Insulin-like Growth Factor (IGF-1) E-peptides modulate cell entry to mature IGF-1 protein. Mol Biol Cel 2009;20:3810-3817.
Choukair D, Hügel U, Sander A, Uhlmann L, Tönshoff B. Inhibition of IGF-1-related intracellular signaling pathways by proinflammatory citokines in growth plate chondrocytes. Ped Res 2014;76(3):245-251.
Liu Q, Guan JZ, Sun Y, Le Z, Zhang P, Yu D, et al. Insulin-like growth factor 1 receptor-mediated cell survival in hypoxia depends on the promotion of autophagy via supression of the PI3K/Akt/mTOR signaling pathway. Mol Med Rep 2017;15:2136-2142.
Tonkin J, Temmerman L, Sampson RD, Gallego-Colon E, Barberi L, Bilbao D, et al. Monocyte/Macrophage-derived IGF-1 orchestrates murine skeletal muscle regeneration and modulates autocrine polarization. Am Soc Gene Cell Ther 2015;23(7):1189-1200.
García-Segura LM, Arévalo MA, Azcoitia I. Interactions of estradiol and insulin-like growth factor-I signaling in the nervous system: New advances. Prog Brain Res 2010;181:251-272.
García-Lacy F, Gutiérrez-Olvera L, Bernad M, Fortier L, Trigo-Tavera FJ, Gómez-Chavarín M, et al. Pharmacokinetic analysis of intraarticular injection of insulin and its effect on IGF-1 expression in synovial fluid of healthy horses. Rev Mex Cienc Pecu 2022;13(2):391-407.
Fortier LA, Balkman CE, Sandell LJ, Ratcliffe A, Nixon A. Insulin-like growth factor-1 gene expression patterns during spontaneous repair of acute articular cartilage injury. J Orth Res 2001;19:720-728.
Frisbie D. Future directions in treatment of joint disease in horses. Vet Clin Equine 2005;21:713-724.
Aguilar IN, Trippel SB, Shuiliang S, Bonassar LJ. Comparison of efficacy of endogenous and exogenous IGF-I in stimulating matrix production and mature chondrocytes. Cartilage 2015;6(4):264-272.
Moyer W, Schumacher J, Schumacher J. A guide to equine joint injection and regional anesthesia. Yardley, PA: Veterinary Learning Systems, USA. 2007.
Verwilghen D, Busoni V, Gangl M, Franck T, Lejeune JP, Vanderheyden L, et al. Relationship between biochemical markers and radiographic scores in the evaluation of the osteoarticular status of Warmblood stallions. Res Vet Sci 2009;87(2):319-328.
Chu CR, Beynnon BD, Buckwalter JA, Garrett WE Jr, Katz JN, Rodeo SA. Closing the gap between benck and nedside research for early arthritis therapies (EARTH): report from the AOOSSM/NIH U-13 Post-joint injury osteoarthritis conference II. Am J Sports Med 2011;39(7):1569-1578.
Bonnevie ED, Delco ML, Fortier LA, Alexander PG, Tuan RS, Bonassar LJ. Characterization of tissue response to impact loads delivered using a hand-held instrument for studying articular cartilage injury. Cartilage 2015;6(4):226-232.
Buzas EI, Gyrgöry B, Nagy G, Falus A, Gay S. Emerging role of extracellular vesicles in inflammatory diseases. Nat Rev Reumatol 2014;10(6):3563-3564.
www.uniprot.com. . https://www.afternic.com/forsale/uniprot.com?utm_source=TDFS&utm_medium=sn_affiliate_click&utm_campaign=TDFS_Affiliate_namefind_direct8&traffic_type=CL3&traffic_id=Namefind.
Phillipou A, Papageorgiou E, Bogdanis G, Halapas A, Sourla A, Maridaki M, et al. Expression fo IGF-1 isoforms after exercise-induced muscle damage in humans: Characterization of the MGF E peptide actions in vitro. In vivo 2009;23:567-576.
Nixon AJ, Brower-Toland BD, Sandell LJ. Primary nucleotide structure of predominant and alternate splice forms of equine insulin-like growth factor I and their gene expression patterns in tissues. AJVR 1999;60(10):1234-1241.
Le, KQ, Prabhakar B, Hong, WJ. et al. Alternative splicing as a biomarker and potential target for drug discovery. Acta Pharmacol Sin 2015;36:1212–1218.
Geneser F. Tejido esquelético. Cap 12, Geneser F. Histología. 3rd ed. Madrid, España: Editorial Médica Panamericana; 2001.
Briggs DT, Sadr KN, Pulido PA, Bugbee WD. The use of osteochondral allograft transplantation for primary treatment of cartilage lesions in the knee. Cartilage 2015;6(4):203-207.
Sherman SL, Khazai RS, James CH, Stoker AM, Flood DL, Cook JL. In vitro toxicity of local anesthetics and corticosteroids on chondrocyte and synoviocyte viability and metabolism. Catilage 2015;6(4):233-240.
Descargas
Publicado
Cómo citar
-
Resumen817
-
PDF 431
-
PDF324
-
Full text 494
Número
Sección
Licencia
Los autores/as que publiquen en la Revista Mexicana de Ciencias Pecuarias aceptan las siguientes condiciones:
De acuerdo con la legislación de derechos de autor, la Revista Mexicana de Ciencias Pecuarias reconoce y respeta el derecho moral de los autores/as, así como la titularidad del derecho patrimonial, el cual será cedido a la revista para su difusión en acceso abierto.

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial-CompartirIgual 4.0 Internacional.