https://doi.org/10.22319/rmcp.v13i1.5943
Article
Antimicrobial residues found in poultry commercialized in retail stores from the Metropolitan Area of Guadalajara, Jalisco
Delia Guillermina González-Aguilar a
Maritza Alejandra Ramírez-López a
Iyari Ximena Uribe-Camberos a
Jeannette Barba-León a*
a Universidad de Guadalajara. Departamento de Salud Pública. Camino Ramón Padilla Sánchez No. 2100 Nextipac, 45200, Zapopan, Jalisco. México.
* Corresponding author: jeannette.barba@academicos.udg.mx
Abstract:
The increased demand to produce large quantities of meat and animal products for human consumption has promoted the indiscriminate use of antimicrobials. The increased use of these substances in the production of poultry, has negative consequences on Public Health due to the fact that the accumulation of antimicrobial residues in the organs and tissues of poultry might reach the consumer. The presence of antimicrobial residues can cause problems of hypersensitivity in humans, or the emergence of antimicrobial resistant pathogens. The purpose of this work was to assess the presence of antimicrobial residues in kidney and muscle tissue of poultry, commercialized in four municipalities of the Metropolitan Area of Guadalajara, Jalisco. The results show that kidney samples had a higher number of positive results compared to muscle tissue. Inhibitors of the folate pathway (sulfamethazine) were the antimicrobials with the highest number of positive results in kidney samples. In contrast, in muscle tissue, β-Lactam (penicillin) were the antimicrobials with the highest number of positive samples. Regarding the analysis of the results by municipalities, it was observed that one of them showed a greater number of positive samples for all the classes of antimicrobials evaluated. This work shows the presence of antimicrobial residues in kidney and muscle tissues of poultry, commercialized in retail sites. Therefore, it is necessary to increase efforts to monitor and control the use of antimicrobial in poultry farms.
Key words: Antibiotic residues, Poultry meat, Jalisco, Retail sale.
Received: 16/02/2021
Accepted: 15/06/2021
Introduction
The poultry industry in Mexico is one of the most productive in terms of meat production(1). The increase on the demand for poultry meat has increased the use of antimicrobials in its production; either to control animal diseases or as growth promoters(2). In Europe, it is estimated that approximately 80 % of chicken poultry receives at least once antimicrobial treatment during their growth, with an average of 172 mg/kg from hatch to slaughter(3).
One of the risks to human health, associated with the use of antimicrobials during intensive poultry production, is the presence of antimicrobial residues in the meat consumed. The presence of antimicrobial residues in the meat can promote health risks in consumers such as toxicity, immunopathological diseases, allergic reactions, carcinogenic effects, among other illnesses(4,5). Additionally, the presence and ingestion of antimicrobial residues, can promote the appearance of drug-resistant pathogens(6). According to the Center for Disease Control and Prevention (CDC), the emergence of antimicrobial-resistant pathogens, such as Campylobacter and Salmonella, is one of the greatest Public Health challenges of this century(6,7).
It has been reported that the abuse of antimicrobials and their inappropriate use, in relation to the dose applied and the period of non-antimicrobials application to the animals before their slaughter (abstinence period), is the main cause of accumulation of antimicrobial residues in the organs and tissues of poultry(4,5). In this regard, antimicrobial residues can be located with different concentrations in several tissues, a situation that depends on the antimicrobial class and its route of administration(8). In addition to the study of the presence of antimicrobial residues in tissues intended for human consumption, several studies use the kidney as a sample matrix, since it is the organ responsible for excreting most drugs(9). Most antimicrobials, such as β-lactams, tetracyclines, streptomycin, sulfamethazine, and chloramphenicol, have been reported to be excreted in the urine either as the parent drugs or as a derived metabolite(10). Therefore, in this study the presence of penicillin, sulfamethazine and streptomycin was analyzed as an indicator of the use of these antimicrobials during the production of the analyzed poultry.
Scientific evidence shows that the classes of antimicrobials commonly used in the poultry industry in the world are aminoglycosides (streptomycin)(11), β-lactams (penicillin)(12), folate pathway antagonists (sulfonamides-sulfamethazine), quinolones (ciprofloxacin) and tetracyclines(13). The toxicity of each one of these classes is different, for example, aminoglycosides are hepatotoxic and ototoxic, Folate pathway inhibitors have teratogenic effects, and can cause urinary tract problems. On the other hand, β-Lactam causes neurotoxicity in cases where kidney function is altered or there are pre-existing brain lesions. Additionally, the three antimicrobial classes cause the disruption of the intestinal flora(5). In Mexico, these three classes are listed as drugs subject to monitoring in poultry(14). Therefore, this study tested the presence of three antimicrobial residues in kidney and muscle tissue of poultry in four municipalities of the Metropolitan Area of Guadalajara (MAG). The antimicrobial residues evaluated in this study are representative of the three antimicrobial classes monitored in Mexico.
Material and methods
Collection of samples
A total of 177 kidneys and 177 muscle tissues, corresponding to the lower back (rump) of an equal number of poultry carcasses, were purchased in retail stores of four municipalities of the MAG. The samples were collected randomly, from February to June 2018. The municipalities tested were Guadalajara (A) with 43 samples assessed for each tissue tested (43 kidney samples and 43 muscle samples), Tlaquepaque (B) with 40 samples, Tonalá (C) with 44 samples and Zapopan (D) with 50 samples. The number of samples per municipality was calculated with Win Episcope v 2.0 software, considering a confidence of 95% and an error of 5%. Kidney and muscle tissue samples were transported in coolers to the Inocuidad de Alimentos Laboratory of the Departamento de Salud Pública of the Centro Universitario de Ciencias Biológicas y Agropecuarias, Universidad de Guadalajara, for analysis.
Sample processing
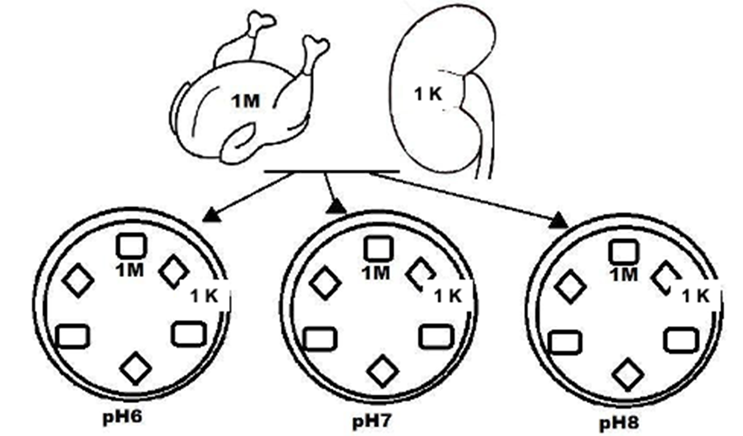
All the kidney and muscle tissue samples collected were dissected in triplicate with a sterile 8 mm punch, under aseptic conditions. The presence of antibiotic residues was assessed by the method of Bacillus subtilis (6 log10 CFU) plate diffusion agar(15,16) using MacConkey agar. The agar was made with three different pH's, where each pH allows testing a different class of antimicrobial. Medium with pH 6 was used in order to test for the presence of β-Lactam residues and was added with penicillin (0.10 I.U.); the medium with pH 7 was used to test for residues of the Folate pathway inhibitors, and it was added with sulfadiazine (0.5 μg); the medium with pH 8 was used to test for aminoglycosides residues, and it was added with streptomycin (0.5 μg). Each one of the samples obtained from kidney and muscle tissue were placed onto plates, corresponding to each one of the antimicrobials assessed, as shown in Figure 1. A 6 mm square of sterile filter paper (Whatman 4) was used as a negative control. The plates were incubated at 37 + 1 °C/24 h. The presence of antimicrobial residues was assessed based on the diameter of the inhibition zone formed in the growth plate of B. subtilis. The measurement was carried out with a vernier ranging from the outer edge of the sample to the end point of the inhibition zone. Inhibition zones equal or greater than 2 mm were considered positive; inhibition zones smaller than 2 mm but equal or greater than 1 mm were considered suspicious and inhibition zones smaller than 1 mm were considered negative(15).
Figure 1: Scheme of the sample’s placement on the antimicrobial plates. M: muscle, K: kidney
Statistical analysis
The data were analyzed with GraphPad Prism 8.4.2. software. Mann Whitney test, Kruskal-Wallis test and post hoc Dunn's multiple comparisons test was used. A P<0.05 was considered a statistically significant difference.
Results
Kidney and muscle tissue samples from 177 chicken carcasses were assessed for antimicrobials residues of penicillin (pH 6), sulfamethazine (pH 7) and streptomycin (pH 8) using B. subtilis plate diffusion agar assay. The 33.3 % (59/177) of samples tested were positive for the presence of any of the antimicrobial residues evaluated, where 22.6 % (40/177) of the samples came from kidney specimens and 10.7 % (19/177) came from muscle tissue (P<0.05) (Table 1). In contrast, 44.1 % (78/177) of the samples were classified as suspicious, where 37.3 % (66/177) corresponded to kidney and 6.8 % (12/177) to muscle tissue (P<0.05). The 40.1 % (71/177) of the kidney samples and the 82.5 % (146/177) of muscle tissue samples were negative. Municipality B was the one with the highest percentage of positive samples reported (18.0 %), followed by municipality D (7.4 %) and municipality A (6.8 %). Regarding the suspicious results, municipality D showed the largest number of samples in this category (22.1 %), followed by municipality B (10.1 %) and municipality A (7.9 %). Municipality C was the one that showed the lowest number of positive (1.2 %) and suspicious samples (4.0 %).
Table 1: Presence of antimicrobial residues in poultry sold in MAG retail stores
Municipality | Samples (n) | Resistant samples (%)* | Suspicious samples (%) | Negative samples (%) | |||
Organ | M | K | M | K | M | K | |
A | 43 | 2.3 | 4.5 | 1.7 | 6.2 | 20.3 | 13.6 |
B | 40 | 5.6 | 12.4 | 1.1 | 9.0 | 15.8 | 1.1 |
C | 44 | 0.6 | 0.6 | 0.0 | 4.0 | 24.3 | 20.3 |
D | 50 | 2.3 | 5.1 | 4.0 | 18.1 | 22.0 | 5.1 |
Total | 177 | ||||||
*The percentage shown was calculated based on the total of muscle tissue (M) and kidney (K) samples analyzed.
The analysis of the positive samples by the type of residues tested, showed a similar number of positive samples in kidneys (P>0.05) (Figure 2). In contrast, penicillin was the residue that showed the highest number of positive samples in muscle (8/177), followed by streptomycin (6/177) and sulfamethazine (5/177) (P>0.05). Regarding the number of suspicious samples, it was observed that kidney was again the organ with the highest number of suspicious samples, penicillin being the residue with the highest number of positive results (30/177) followed by streptomycin (21/177) and sulfamethazine (15/177). Interestingly, muscle samples reported fewer suspicious results for sulfamethazine (6/177), penicillin (4/177) and streptomycin (2/177). The statistical analysis of suspicious samples showed statistically significant differences between the type of samples (K vs M) (P<0.05). In contrast, no statistically significant difference was found by class of antimicrobial residue (P>0.05).
Figure 2: Positive and suspicious samples by antimicrobial assessed
The graph shows in white the number of positive samples for each of the antimicrobials tested. The number of suspicious samples per antimicrobial tested is shown in gray.
ABCDE Different capital letters indicate statistically significant differences (P<0.05).
The analysis of the antimicrobial residues results by locality showed that municipality B is the one with the highest number of positive samples for the three residues evaluated in both types of evaluated samples (K and M). Statistical analysis showed statistically significant differences for the number of positive samples for ampicillin residue in kidney tissue between municipalities B, C and D (P<0.05). For sulfamethazine, statistically significant differences were observed between municipalities B and C (P<0.05). Finally, for streptomycin residue, the differences were observed in municipalities A, B and C (P<0.05) (Figure 3a). Regarding the number of suspicious samples in kidney, it was found that municipality D was the one with the highest number of samples reported for the three residues evaluated compared with the rest of the municipalities (P<0.05).
A similar pattern was observed for muscle tissue, where municipality B was the one with the highest number of positive samples reporting the three residues evaluated (P>0.05) and municipality D the one with the highest number of suspicious samples for sulfamethazine and streptomycin residues (P>0.05) (Figure 3b). Interestingly, in muscle tissues, municipality C did not report positive or suspicious samples for sulfamethazine and streptomycin residues and only reported one positive sample for ampicillin residue.
Figure 3: Positive and suspicious samples evaluated by antimicrobial and municipality
The graph shows in white the number of positive samples for each of the antimicrobials tested. The number of suspicious samples per antimicrobial tested is shown in gray.
* indicate statistically significant differences (P<0.05), between the bars indicated by the arrowheads.
Discussion
Several investigations have reported the implications of the presence of antimicrobial residues in the tissues of animals destined for human consumption, both in human health as well as in the development of drug resistance in zoonotic pathogens. Currently, the World Health Organization indicates that, in order to prevent and control the spread of antimicrobial resistance, the agricultural sector should administer antibiotics to animals only under veterinary supervision, avoiding the use of antibiotics as growth promoters or to prevent diseases in healthy animals. The use of vaccines is recommended in animals destined for consumption, in order to reduce the need for the use of antibiotics. Likewise, it is recommended to improve hygiene at control points throughout the production chain(17). It has been reported that the emergence of zoonotic pathogens resistant to multiple classes of antimicrobials can spread from the animal to food, causing difficult-to-treat food-borne diseases in humans, resulting in a significant health, medical and socioeconomic impact(18).
The inappropriate and excessive use of antimicrobials in poultry, evidenced by the presence of antimicrobial residues in organs such as the kidney and/or tissues, promotes either that the microbiota of poultry, or the zoonotic pathogens present within the animals, obtain, and transmit antimicrobial resistance genes by selection pressure. In consumers, the presence of antimicrobial residues in food can affect their health in two main ways: (1) promoting allergic and toxic reactions, due to prolonged exposure to low levels of residues or (2) due to a possible appearance and spread of antimicrobials resistance in the host microbiota or in pathogens that cause a difficult to treat infection(19). In this regard, in Vietnam found a relationship between the antimicrobial residues present in poultry meat from wet markets and supermarkets and non-typhoid Salmonella (NTS) strains obtained from the same samples. The study consisted of the analysis of 119 samples, where the presence of 10 antimicrobial residues was found. The classes of Folate pathway inhibitors, tetracyclines and macrolides were the ones that showed the highest number of NTS resistance. The prevalence of NTS was 71.8 %, the most common serotypes being Kentucky, Corvallis, Agona and ST2024. From the Salmonella isolates recovered, it was observed that bacterial resistance and the presence of antimicrobial residues in poultry meat corresponded to the classes of tetracyclines and Folate pathway inhibitors. The results shown in the Vietnam study emphasize that there is a correlation between the presence of antimicrobial residues in poultry tissues and the isolation of resistant pathogenic bacteria(20).
This study shows the presence of residues in kidney and muscle tissues to three different classes of antimicrobials, β-Lactam (penicillin), Folate pathway inhibitors (sulfamethazine) and Aminoglycosides (streptomycin), from poultry commercialized in four municipalities of the MAG (Table 1). The percentage of positive samples (33.3 %) found in this study is higher than that found in raw beef in Erbil, Iraq (10.8 %)(21) and in poultry in Shanghai, China (22.2 %)(19) and is lower than that reported in poultry food (47.3 %) in Fujian, China(22). The results obtained in this study compared to those obtained in Iraq and China show that despite the existence of regulations and antimicrobial residue control programs, their surveillance and control is not efficient. Either due to lack of infrastructure and diagnostic tests to identify antimicrobial residues in the organs and tissues of poultry or due to lack of experience to detect these residues.
The results obtained here showed that the Folate pathway inhibitors were the ones with the highest number of positive samples reported in kidney, followed by β-Lactam and Aminoglycosides residues. In contrast, in muscle samples, the residue that reported the highest number of positive samples was β-Lactam, followed by Aminoglycosides and Folate pathway inhibitors. The observed results contrast with the results reported in China, where Tetracycline is the antimicrobial residue with the highest incidence in poultry(22). Likewise, the results of this study are in agreement with those reported in Pakistan and Nigeria, where the residues of Folate pathway inhibitors (sulfonamides) were the antibiotic class with the highest incidence in poultry(23,24). It appears that the presence of antimicrobial residues belonging to the classes of Folate pathway inhibitors and Tetracyclines is dependent on the antimicrobial frequently used in each country. The presence of antimicrobial residues in México, China and Africa shows the lack of economic resources that would allow the acquisition of supplies and technical training to carry out an adequate antimicrobial residue control program. In Mexico, although there are government regulations for antimicrobial residue control programs, these are only carried out in slaughter plants whose product is destined for export products, neglecting local consumption. On the other hand, studies have reported that positive kidney samples for antimicrobial residues are higher than those found in muscle, due to the fact that the abstinence period in the application of the antimicrobial is reflected more quickly in the muscle than in the kidney(25). For this reason, a positive result in kidney should not be considered an indicator of muscle tissue quality(25). In this regard, it has been reported that aminoglycoside residues remain in the kidney for prolonged periods of time, even months, due to their affinity to the renal cortex(26).
Regarding the results of the number of samples positive to multiple classes of antibiotics, such as those observed in municipalities A, B and D, the results obtained are similar to those reported in China and the Netherlands(22,27). In China, it was reported that 28.8 % of the positive samples were positive for two or more antibiotic residues, where the tetracycline residue was the most frequently found, followed by the residues of Folate pathway inhibitors(22). Additionally, the differences observed in this study regarding findings obtained for municipalities A, B and D are possibly due to the origin of the broilers that process each slaughter plant. The results obtained suggest that the animals slaughtered in the plants of municipalities A and D come from farms that carry out some operational control program. In contrast, the results observed in municipality B suggest that broilers come from farms where there is no adequate control in the administration and suspension of antimicrobial treatments prior to slaughter.
Conclusions and implications
The results obtained show that the analyzed poultry reach the slaughter plants with the presence of antimicrobial residues in the kidney and muscle. The presence of antimicrobials analyzed in broilers suggests abuse in their administration. Since there are not traceability studies of the animals received in each slaughterhouse analyzed in this study, it is necessary to carry out studies that monitor the different classes of antimicrobials and doses administered to poultry. However, the results of this study only reflect a local problem in Jalisco, Mexico and the data in this study do not necessarily reflect the situation in the country. Likewise, the lack of human risk assessment studies within Mexican populations, makes it difficult to analyze the risk of exposure to antimicrobials, due to the ingestion of poultry meat, both locally and nationally.
Acknowledgements
The authors thank Kevin Brian Magallon Carrizales and Sergio Arturo Cordova Ramírez for his technical support.
Declaration of competing interest
All authors declare that there are no conflicts of interest.
Literature cited: